Paper Records vs. LIMS: Choosing the Right Data Management Strategy for Your Lab
Author

Neerav Singh
Technical Product Specialist

Author

Neerav Singh
Technical Product Specialist
Reading Time
3 min read
Paper Records vs. LIMS: Choosing the Right Data Management Strategy for Your Lab
For decades, paper-based record keeping was the backbone of laboratory operations. Binders stacked on shelves, handwritten test logs, manually updated calibration sheets; it was the norm. But as product testing grows more complex and compliance demands intensify, the question is no longer whether to modernize your lab data management, but how fast you can afford to wait.
This blog breaks down the real differences between paper records and a Laboratory Information Management System (LIMS) and helps you decide which data management strategy is right for your testing environment.
The Case for Paper Records and Why It's Fading
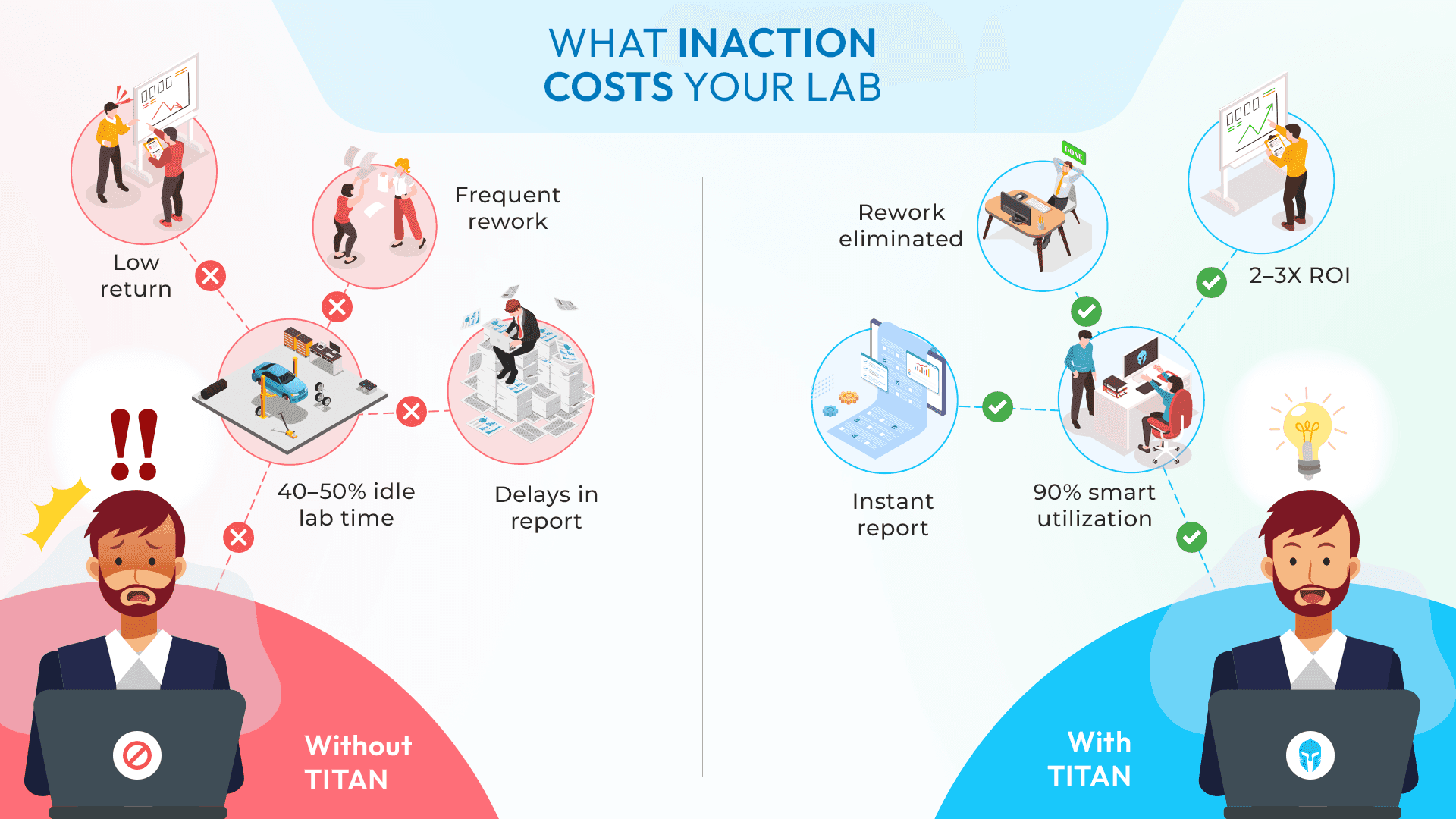
Paper records are simple. No software license, no onboarding curve, no IT dependency. For small, low-volume labs with infrequent testing cycles, manual documentation can feel entirely manageable. But the moment your test volume scales, the cracks begin to show.
Paper-based systems suffer from a fundamental problem: they are static. Once data is written down, it cannot be searched, filtered, cross-referenced or analyzed without someone manually doing it. In environments where test lifecycle management, equipment calibration logs and regulatory reporting are all operating simultaneously, this creates serious bottlenecks.

Data retrieval that should take seconds can take hours. And the risks go beyond inefficiency. Human error remains one of the leading contributors to quality issues, particularly in environments dependent on manual documentation. In validation workflows, even small inconsistencies in recorded test conditions or results can lead to rework, failed audits or incorrect conclusions.
At the same time, the lack of structured traceability introduces compliance risk. Inadequate data management significantly increases the likelihood of non-conformance in manufacturing and testing systems. Standards such as ISO 9001 explicitly require controlled documentation and traceable records, which paper-based systems struggle to maintain at scale.
The impact is not just operational but financial. The cost of poor quality, driven by errors, rework and inefficiencies, can account for up to 15–20% of revenue in many organizations
From a regulatory compliance standpoint, paper trails are notoriously difficult to audit. Tracing a single test result back through its requirements, test conditions, equipment state and personnel sign-offs requires piecing together records from multiple binders, a process prone to gaps, inconsistencies and non-conformances.
What Is a LIMS And What Does It Actually Solve?
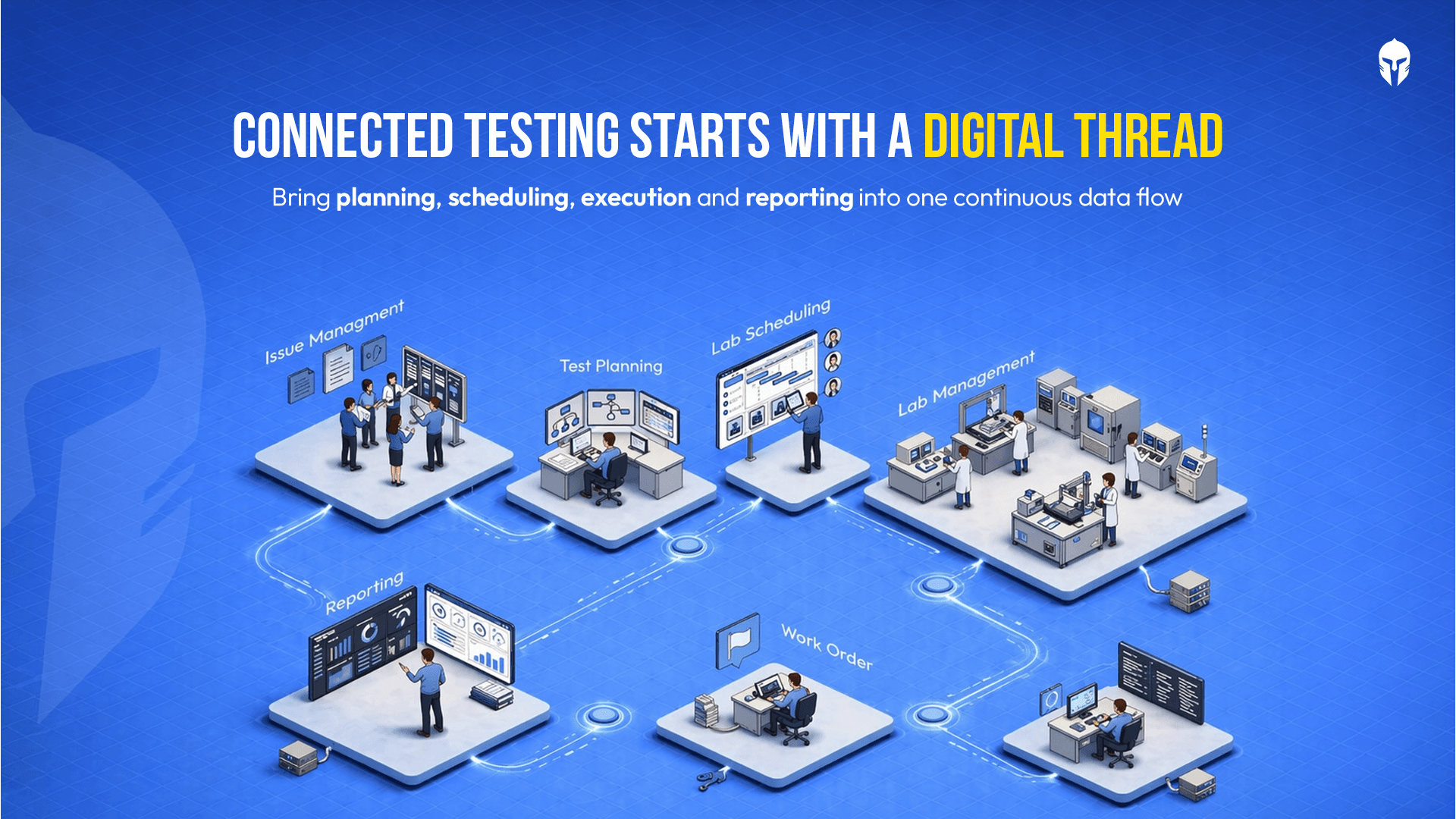
A Laboratory Information Management System (LIMS) is a software platform designed to digitize, centralize and automate the flow of data. It connects your test requests, sample tracking, equipment management, results and reporting into a single, searchable system.
The key advantages of a LIMS over paper records include:
- Data Integrity and Audit Trails.
Every entry in a LIMS is timestamped, user-attributed, and version-controlled. Any change ,intentional or accidental is logged. This is critical for compliance, ISO/IEC 17025 accreditation and any industry requiring a verifiable chain of custody for test results. - Real-Time Data Access.
With a LIMS, test data is available the moment it's entered accessible across teams, shifts and locations. There's no waiting for a physical report to travel from the lab floor to an engineer's desk. - Traceability Across the Test Lifecycle.
A robust LIMS stores data along with connecting them. From the original requirement, through the test plan, the equipment used, the personnel involved and the outcome, end-to-end traceability is built in. This is where modern platforms that cover the full test lifecycle deliver their biggest value. - Reduction in Human Error.
Manual data entry is a known source of errors in lab environments. Automated data capture, dropdown validation and system-enforced workflows significantly reduce the risk of transcription errors, missed calibration checks or mislabeled samples.
The Drawbacks of Staying on Paper
When labs calculate the cost of paper records, they often only count the cost of the paper itself. The real cost is invisible until something goes wrong.
Consider: a failed regulatory audit due to incomplete test documentation. A product recall triggered by a test result that couldn't be traced back to its source. A duplicated test run because no one could locate the original data. These are not hypothetical scenarios, they happen often in labs that haven't modernized their data management infrastructure.
Beyond risk, there's the daily operational drag. Engineers spending hours searching for historical records instead of running new tests. Lab managers unable to see real-time equipment availability or test scheduling conflicts. Reporting that takes days to compile manually, instead of being generated automatically.
Multiple researches consistently depict that labs transitioning from paper to digital LIMS solutions report significant gains, 55% faster turnaround times, improved cross-team collaboration and measurable reductions in quality issues. Platforms like TITAN TLM, built specifically for R&D and validation labs, demonstrate these outcomes with teams across automotive, aviation, marine, consumer electronics testing and beyond.
When Should You Make the Switch?
Not every lab needs an enterprise LIMS on day one. But there are clear signals that paper records and spreadsheets are no longer sufficient:
Your test volume has grown to the point where retrieval and reporting consume significant lab hours. Your lab operates across multiple teams or locations and needs centralized data visibility. You are preparing for or maintaining ISO/IEC 17025 or any industry-specific accreditation. You are managing complex equipment calibration schedules, maintenance histories and asset tracking. Your stakeholders or clients require structured, traceable test reports on tight timelines.
If any of these describe your environment, the transition from paper to a digital lab management system is no more a luxury.
Choosing the Right LIMS for Your Lab
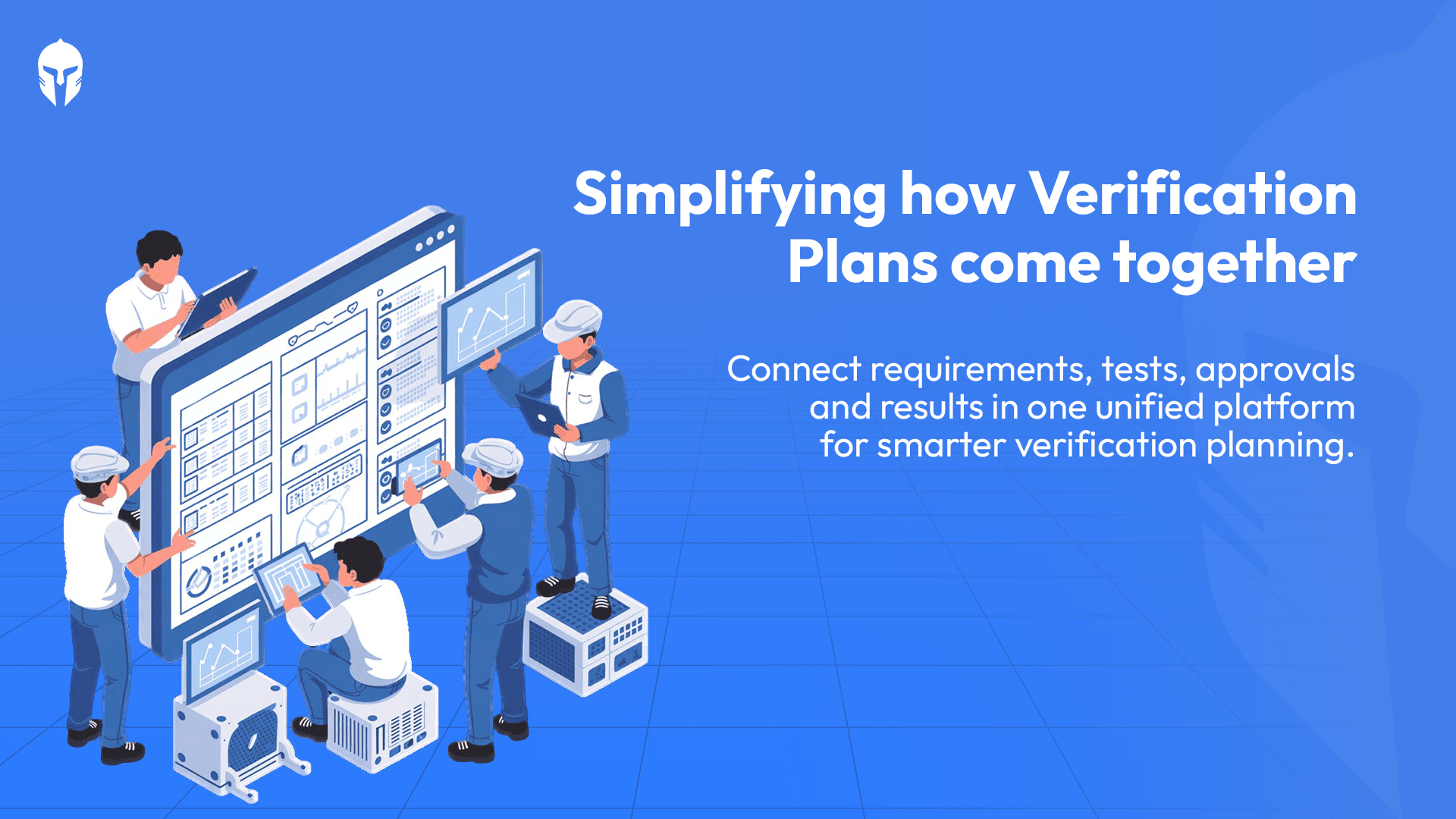
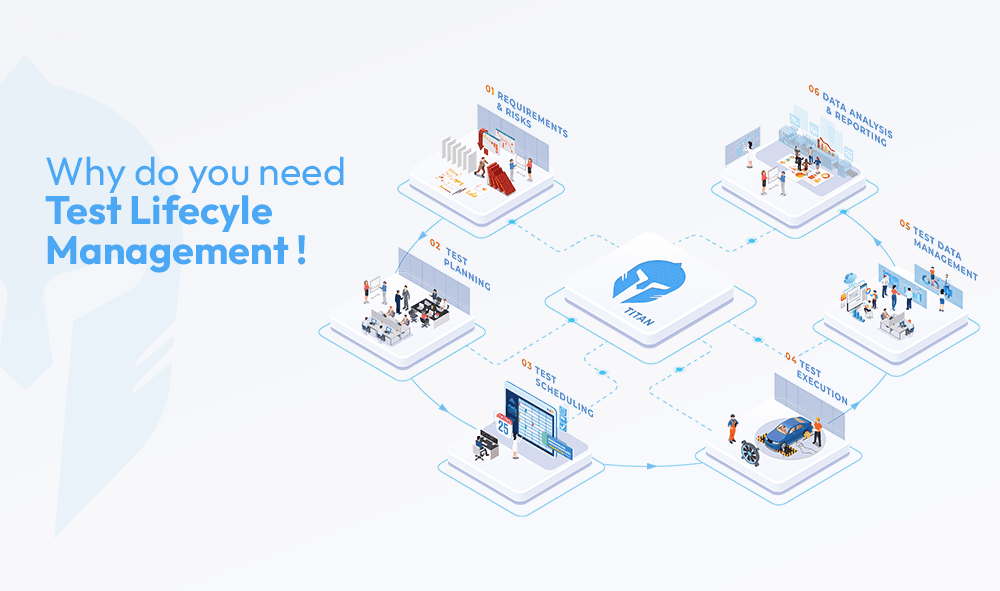
Not all LIMS platforms are built the same. When evaluating options, look for a solution that covers the full test lifecycle not just data capture, scheduling, work order management, equipment tracking and reporting in one unified platform.
A fragmented tool landscape, one system for scheduling, another for data, another for reporting recreates many of the same problems that paper records cause. The goal is unified and centralized lab data management that gives every stakeholder a single source of truth.
The right platform should also be configurable to your workflows, scalable as your testing operations grow and capable of integrating with your existing tools through APIs or pre-built connectors. Modular deployment matters too, you shouldn't have to overhaul everything at once to start seeing value.
Why TITAN Works Better Than Traditional LIMS
Most LIMS platforms focus on storing and managing lab data. TITAN goes further by structuring how testing operations actually run.
Unlike conventional LIMS tools that operate as standalone data repositories, TITAN is built as a test lifecycle management platform. It connects every stage of testing into a single system, reducing fragmentation and improving visibility across teams.
Here’s where TITAN stands out:
- End-to-End Test Lifecycle Coverage
TITAN connects test requests, planning, scheduling, execution and reporting in one unified platform. There is no need to switch between multiple tools. - Built for Test Scheduling
Many LIMS tools do not handle scheduling well. TITAN provides clear visibility into equipment availability, test slots and resource allocation, helping labs reduce idle time and avoid conflicts. - Stronger Traceability Across Workflows
Every test is linked to its requirements, equipment, approvals and results. This makes audits easier and ensures complete data continuity. - Configurable Workflows for Lab Operations
TITAN adapts to how your lab operates. Approval flows, test processes, and data structures can be configured without complex customization. - Real-Time Operational Visibility
Lab managers and engineers can track test progress, equipment usage, and bottlenecks without relying on manual updates or follow-ups. - Designed for R&D and Validation Labs
TITAN is purpose-built for environments where testing is iterative, multi-stage, and dependent on coordination across teams.
Instead of only digitizing records, TITAN improves how testing work moves through your lab.
To Summarize
Paper records served their purpose but the demands of modern product testing have outgrown them. LIMS adoption is no longer just about going digital. All labs are chasing to build a lab operation that is traceable, auditable, collaborative and swift enough to support today's development cycles.
The decision between paper and a digitized laboratory data management system ultimately comes down to one question: can your current approach scale with the complexity and compliance expectations your lab will face in the next three years?
If the answer is uncertain, it's time to explore what a modern test lifecycle management platform can do for your team.
To see how integrated test lifecycle management transforms lab data operations, visit testlifecycle.com
Explore the full suite of lab, data, and test management capabilities.
Built to simplify complex lab operations and bring clarity to every test.