Implementing ALCOA in Your Lab: Gaps, Best Practices and What Good Looks Like
Author

Neerav Singh
Technical Product Specialist

Author

Neerav Singh
Technical Product Specialist
Reading Time
3 min read
Implementing ALCOA in Your Lab: Gaps, Best Practices and What Good Looks Like

In the previous blog, we covered what the ALCOA principles are and why they apply to engineering and testing laboratories every bit as much as they do to pharmaceutical manufacturing. Understanding the framework is the necessary first step. The harder question and the more operationally urgent one is what ALCOA compliance looks like in a real lab and where the gaps most commonly appear. This blog focuses on the practical side: the three most prevalent failure points in engineering testing environments, the best practices that close them and the broader organizational case for applying ALCOA beyond any single regulatory requirement.
ALCOA in Engineering Testing: The Practical Gap
Understanding the ALCOA data integrity framework for engineering testing at a conceptual level is straightforward. The more difficult question is what it looks like in practice and where the gaps most commonly appear in real testing environments.
Gap 1: Contemporaneous Recording
The most prevalent gap is in contemporaneous recording. Engineering test programs move quickly. Engineers manage multiple test events, equipment handovers and data streams simultaneously. When documentation is treated as something that happens after testing rather than as part of it the contemporaneous principle is compromised at scale. The records may be accurate, but they are reconstructions. And reconstructions introduce ambiguity that becomes significant under audit or investigation.
Gap 2: Original Data Preservation
The second most common gap is in original data preservation. In laboratories where measurement data flows from instruments through processing software into spreadsheets and eventually into reports, each transition represents a potential break in the chain of original data integrity. Unless the system is designed to preserve the original capture and maintain an unbroken link to all subsequent records, the "original" principle is practically unenforceable.
Gap 3: Audit Trail and Traceability
Audit trail and traceability in testing labs is the third area where gaps are common. An audit trail is not just a log of who did what. It is a reconstruction capability, the ability to take any result in any report and trace it back through the data chain to the original source, the equipment that produced it, the calibration state of that equipment at the time and the procedure under which it was run.
Manual systems cannot reliably provide this. The reconstruction takes time, depends on the continuity of individual knowledge and produces outcomes that are difficult to verify. The importance of ALCOA in management for testing laboratories ultimately comes down to this reconstruction capability. Organizations that have it can respond to audit requests, investigate anomalies, and defend their results with confidence. Organizations that do not are vulnerable every time that capability is tested.
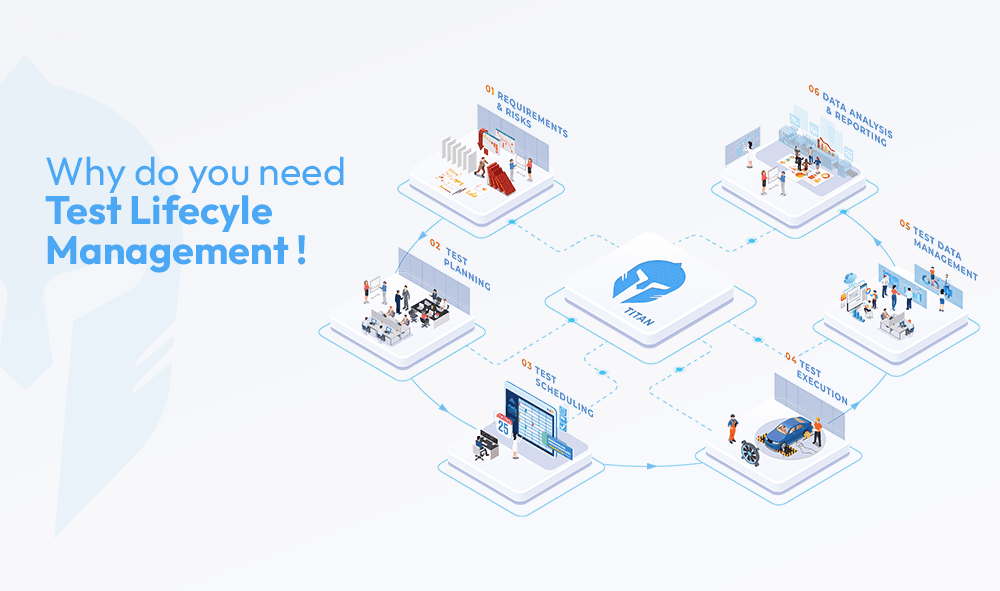
Best Practices for ALCOA-Aligned Data Management in Testing Laboratories
The transition from awareness to implementation requires both cultural and structural changes. ALCOA-aligned data management shares several consistent characteristics across well-run testing environments.
Capture data at the point of activity. Systems and workflows should be designed so that recording is part of the activity, not a separate step that follows it. When data entry happens in the same interface where the test is being managed, contemporaneous recording becomes the default rather than the exception.
Preserve original records by design. Data management systems should store original captures separately from processed or reported outputs, with a traceable link between them. Overwriting original data even inadvertently should be technically prevented rather than procedurally discouraged.
Make attribution automatic. Every data entry, every approval, every modification should be attributed automatically to the authenticated user who performed it. Systems that require manual attribution create both administrative overhead and attribution gaps whenever the process is not followed consistently.
Maintain a complete and searchable audit trail. Every change to a record should be logged not just the final state, but the history of how it arrived at that state. Deletions, corrections and approvals should all appear in the audit trail with timestamps and user identities.
Connect records across the test lifecycle. Data integrity is not just about individual records. It is about the relationships between them between a test result and the procedure that governed it, between a measurement and the calibrated equipment that produced it, between a report and the raw data that underlies it. Systems that manage these connections structurally, rather than relying on documentation conventions, provide a more reliable basis for traceability.
Platforms designed for test lifecycle management such as TITAN are built around these requirements. They bring together test planning, execution, data capture, calibration tracking and reporting in a single connected environment where ALCOA-aligned management is an operational reality rather than a compliance aspiration.
ALCOA Principles Beyond Pharma: The Broader Implication
The pressure on data integrity standards is increasing across sectors. Regulatory frameworks in automotive, aviation and consumer electronics are becoming more stringent. Customer audit requirements are becoming more specific about data traceability and record integrity. ISO 17025 accreditation, where it is held or sought, imposes requirements that align closely with ALCOA whether the framework is referenced explicitly or not.
The ALCOA principles represent not an imported compliance burden but a practical standard for what reliable data management looks like in any high-stakes testing environment. The organizations that apply them are better positioned for audits, more confident in their results and more operationally resilient when things do not go according to plan.
The principles themselves have not changed. Only the context in which they are being applied has widened.
To Summarize It All
The infrastructure to implement ALCOA well now exists. The case for doing so is not limited to any one industry. And the cost of not doing it is becoming harder to justify regardless of where the pressure to act originates.
Want to see how TITAN supports ALCOA compliance in testing labs?
Ensure data integrity, traceability, and audit-ready records across your entire test lifecycle.